THE
SEA'S MISSING SALT:
A
DILEMMA FOR EVOLUTIONISTS
Steven
A. Austin, Ph.D
D. Russell Humphreys, Ph.D.1
Institute for Creation Research Santee,
California 92071
From "Proceedings of the Second
International Conference on Creationism," 1Dr.
Humphreys is Adjunct Professor at ICR and a physicist at Sandia National
Laboratories, Box 5800, Div. 1261, Albuquerque, NM 87185. The Laboratories have
not supported this work. Return To ABSTRACT The known and conjectured processes which deliver and remove dissolved
sodium (Na+). to and
from the ocean are inventoried. Only 27% of the present Na+ delivered
to the ocean can be accounted for by known removal processes. This indicates
that the Na+ concentration
of the ocean is not today in "steady state" as supposed by
evolutionists, but is increasing with time. The present rate of increase (about
3 x 10" kg/yr) cannot be accommodated into evolutionary models assuming
cyclic or episodic removal of input Na+ and a
3-billion-year-old ocean. The enormous imbalance shows that the sea should
contain much more salt than it does today if the evolutionary model were true.
A differential equation containing minimum input rates and maximum output rates
allows a maximum age of the ocean of 62 million years to be calculated. The data can be
accommodated well into a creationist model. INTRODUCTION Sodium
is the most common dissolved metal in the ocean. It exists in seawater as a
positively charged ion. Sodium ions (Na+) form the
primary salt of the sea along with negatively charged chloride ions (Cl-).
The extreme solubility is caused by the cation's small size (ionic radius is
0.97 Å) and small
charge (single positive charge), which allows Na+ ions to escape most
geochemical processes which remove larger ions with the same or greater charge. The
worldwide delivery of Na+ to the
ocean by rivers has been recognized by scientists for hundreds of years. Almost
three hundred years ago Edmund Halley [1] recognized that salt cannot easily
leave the ocean and suggested that the age of the ocean might be established
from knowledge of how much salt enters it year by year from rivers. Nearly one
hundred years ago John Joly [2] measured the amount of Na+ dissolved in river water and estimated with extraordinary
accuracy the global yearly input of Na+ to the
ocean. Joly said it would take 80 to 90 million years for the sea to accumulate
its present amount of Na+, if it
did so at a constant rate and had none in the beginning. That calculation was
accepted by many scientists as giving the age of the earth. By 1930
radioactive dating methods had been developed which indicated that the age of
the earth was longer than anyone had anticipated. Many scientists became convinced
that the earth and the ocean are billions of years old. These scientists could
no longer endorse Joly's method which they recognized "...leads to the
spuriously low geochemical age"[3]. F. W. Clarke, V. M. Goldschmidt, and
W. W. Rubey [4] were among many who conjectured that Na+ is removed from the ocean about as fast as it
enters, causing the amount of Na+ in the
ocean to remain roughly constant with time. C. B. Gregor reaffirmed their
belief recently: "If magma kept the crust built up against the ravages of
erosion and the waste products accumulated in the sea, at present rates of
influx the ocean basins should long ago have been choked with sediment and salt
.... salt must somehow leave the ocean."[5] Those who endorse a 4.5
billion year old earth agree that Joly's 80 to 90 million years is not the age,
but the "residence time" for Na+,
that is, the average length of time the ion would survive in the ocean
before being removed. The interpretation that the ocean is in "steady
state" with respect to Na+
was brought to creationists' attention again by Howard J. Van Till, Davis
A. Young and Clarence Menninga in Science
Held Hostage[6]. They endorse radioactive isotope
dating and insist that evolutionists are correct when they suppose that the
rate of addition of Na+ to the
ocean is balanced by removal processes of equal magnitude. The "residence
time" for Na+,
they assert, provides no means for establishing an age for the ocean.
They affirm, "The 4.5 billion year chronology of earth history is in no way
weakened or disqualified by an appeal to the salt content of the terrestrial
oceans."[7] But where is the empirical evidence supporting the
"steady state" model? Is there sufficient reason, apart from
evolutionary assumption, to dismiss Joly's geochemical age for the ocean? Van
Till, Young and Menninga do not present the evidence, but simply endorse the
model supposed by earlier evolutionists. The steady-state hypothesis cannot be tested directly,
because, even if the ocean is not in steady state, the change in Na+ concentration of seawater during
recent times would be too small to be measurable. But there is an indirect test
for the hypothesis; we can compare measured input rates with all known or
conjectured output rates. If outputs are considerably lower than inputs at
present, then the sea cannot be be in steady state. If that condition is likely
to have persisted for the history of the ocean, there is strong reason to doubt
that the sea is billions of years old. Thus, we will examine input and output
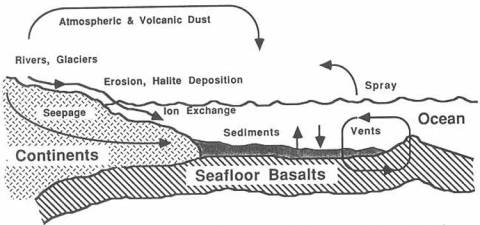
rates carefully. Atmospheric & Volcanic Dust Figure 1: Diagram illustrating the ocean's major inputs and outputs of Na+. Figure 1 illustrates inputs and outputs. We define the
system in question as being the seawater in the ocean basins, not including
water trapped in sea-floor sediments. First, we list all the known or
conjectured inputs and outputs and try to quantify them with the latest data
from the scientific literature. Then, we specify their past behavior in the
evolutionary model. Next, we calculate the maximum possible age of the ocean on
the basis of the evolutionary model, in order to show the inconsistency of that
model. Last, we indicate the concordance of the data with a creation model, and
offer a challenge to evolutionists and old-earth creationists. PRESENT INPUTS OF Na+ TO THE OCEAN Let us define Ai
as the mass per unit time of Na+
delivered to the ocean by the ith source. What follows is a
compilation of eleven major natural Na+
inputs to the ocean. Most considerations of the Na+ cycle for the oceans only
take account of the first three inputs listed (rivers), but we list in Table 1
and below eight additional sources which cannot be neglected. The most thoroughly investigated process delivering Na+ to the ocean is rivers.
The most recent global survey by the French geochemist Michel Meybeck gives
both the total discharge and average Na+
concentration of rivers. The total river discharge to the ocean is 3.74x
1016 L/yr, and the globally averaged Na+ concentration of rivers after man-made pollution is
removed is 5.15 mg/L [8]. These numbers allow the global river input of natural
dissolved Na+ to the
ocean to be calculated as 1.92 x 1011 kg/yr [9]. According to
Meybeck, there are three major sources for Na+ in river water: (1) sea spray, (2) chemical
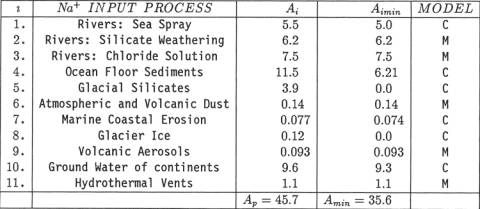
weathering of silicate minerals, and (3) solution of chloride minerals. Table 1: Inputs of Na+ to the world ocean. Units are in 1010 kg/yr.
Present inputs are listed in column headed Ai.
Minimum past inputs are listed in column headed Aimin.
Models for estimating Aimin, are denoted "M" for "Modern Earth
Model" and "C" for "Cretaceous Earth
Model". Al. Rivers: Sea Spray Component. Spray from ocean waves causes droplets
of seawater to evaporate leaving halite aerosol in the atmosphere. A large part
of the aerosol is deposited by rain and snow on the continents. The component
of Na+ in river water derived
from sea spray was estimated by study of the Na+
concentrations of numerous rain samples and the total discharge of rivers
to the ocean. According to Meybeck [9], sea-spray-derived Na+ in river water is 5.5x 1010 kg/yr, which
is 29% of the total yearly river flux of Na+. Thus, A1 = 5.5 x 1010 kg/yr. A2. Rivers: Silicate Weathering
Component. Chemical weathering of the continents releases Na+ from silicate minerals (especially feldspars and
clays) the major part of which finds its way to rivers and enters the ocean.
Analysis of the mineral breakdown of dissolved ions in river water by Meybeck [10]
indicates that 32% of the
total Na+ is derived from
weathering of continental silicate minerals. Thus, A2=6.2x1010 kg/yr. A3. Rivers:
Chloride Solution Component. A small area of the continents (approx. 1.3% of area) has outcrops of chloride and sulfate minerals the
principal mineral of which is halite (NaCl). These
are extremely soluble in water. Using the mineral breakdown analysis of
dissolved ions in river water, Meybeck [10] calculated that 39% of modern river Na+ is
derived from solution of chlorides. Thus, A3 = 7.5 x 1010 kg/yr. The sum A1 + A2 + A3 =
1.92 x 1011 kg/yr,
which is the total global river flux of Na+. A4. Ocean Floor
Sediments. Detailed studies of the Na+
concentrations of pore waters of ocean floor sediments by Sayles [11] show
that their pore waters are enriched in Na+
relative to sea water. The increase in Na+
of pore waters with depth within ocean sediments describes a
concentration gradient which requires diffusion of Na+
from ocean sediments into the ocean. Pore waters of ocean sediments show
decreasing concentrations of K+ and Mg+2 with
depth. This data requires sediments to absorb K+ and Mg+2 from
seawater [11]. A good explanation offered for this data is that most Na+ is released from clays during prolonged burial as K+ or Mg+2 is absorbed in its place.
K+ would be absorbed during
prolonged burial of clays because it has larger ionic radius than Na+. Similarly, Mg+2 would be absorbed by
clays during prolonged burial because of its divalent charge, twice that of Na+. The ocean sediment pore water data for the Atlantic Ocean
assembled by Sayles is representative of other oceans allowing the global Na+ flux out of ocean sediments to be calculated. The
estimate of Sayles [11] is slightly adjusted for charge balance and yields A4 = 1.15x 1011 kg/yr
[12]. This large input to the ocean is 60% of the total river input. A5. Finely
Pulverized Glacial Silicates. Glaciers produce very
finely ground rock flour and, as illustrated by Antarctica and Greenland, add the
minutely pulverized material directly to the ocean. This fine rock flour is
dominated by silicate minerals which weather rapidly when added to the ocean.
Schultz and Turekian [13] describe the silica enriched deep ocean waters surrounding Antarctica and the evidence that about 1.4 x 1012 kg (64%) of the
continent's glacial-marine sediments dissolve in sea water before being buried
on the sea floor. If we assume that the glacial-marine sediment of Antarctica
has 2.4% by weight Na+ (the composition of the
"average igneous rock"), the dissolved silicates add 3.4x 1010 kg of Na+
to the ocean each year. The continent of Antarctica comprises 86% of our
planet's continentally glaciated area and indicates that the global
glacial-marine Na+ flux is AS = 3.9 x
1010 kg/yr.
This is 20% of the total river input. A6. Atmospheric
and Volcanic Dust. A
considerable quantity of the dust removed from the continents by wind is added
to the ocean. The fine dust is largely silicate minerals, a major part of which
dissolve in the sea. According to Garrels and Mackenzie [14], 5 x 1010 kg of
atmospheric dust are added yearly to the ocean basins. This is equivalent to an
average of 140 kg of dust per km2 of ocean each year,
and agrees with dust fluxes collected over the ocean [15]. Mackenzie and Wollast [16] add to this total 4 x 1010 kg of
volcanic dust each year. Assuming that atmospheric and volcanic dust is 2.4% by
weight Na+ and that 64% of it dissolves, A6=1.4x109 kg/yr. This is 1% of the total river input. A7.
Marine Coastal Erosion. The direct attack of ocean waves along the
coast erodes considerable amounts of sand, silt and clay. Most of the finest
particles produced are silicates which have not been rinsed by the fresh water
of rivers and remain very reactive with the ocean. According to Garrels and
Mackenzie [14], 2.3 x 1011 kg of material is added to the ocean yearly by
marine erosion. Assuming that this debris is 0.67% by weight Na+ (the
concentration in the "average sedimentary rock") and that 50% of it dissolves
in the ocean, A7 =
7.7 x 108 kg/yr of
Na+ from marine erosion. This is less than 1% of the total river input. A8. Glacier
Ice. Snow has small quantities of Na+
derived from halite aerosols of the atmosphere. Melting of glacier ice directly
in the sea adds small quantities of Na+ to the sea. Meybeck
[17] lists this value, A8 = 1.2 x 109 kg/yr.
This is less than 1% of the total river input. A9. Volcanic
Aerosols. Dissolved in the steam that continental volcanoes
deliver to the earth's surface are small quantities of Na+. Meybeck [17]
calculated the mass delivered from airborne volcanic aerosols to the ocean. He
obtained A9 = 9.3 x 108. This is
less than 1% of the total river input. A10. Ground Water
Seepage. Geologists have suspected for more than 100 years that
water seeps through the continents and issues forth on the floor of the ocean.
This was not proven until recently when drilling of sediments of the
continental shelves revealed fresh water moving seaward through the sedimentary
layers. The dissolved solids of ground waters can be measured in parts per
thousand (ppt) and vary significantly from as fresh as rain water (0.0001 ppt)
to extremely saline brine (over 250 ppt).
For this reason we have great difficulty in estimating their average Na+
content. Most near surface ground
waters have more dissolved solids than river waters (rivers average 0.13 ppt).
Deep ground waters often are saline brines as an example from the southeastern
United States illustrates. Strata at depth within the Florida-Bahama Platform
are filled with dense brine (over 200 ppt)
that seeps from the platform onto the floors of the Atlantic Ocean and the Gulf
of Mexico [18]. According to Paull and Neumann [18], major brine seepage causes
significant solution of limestone and is believed to be a major cause of the
steep slopes at the margins of the platform. The salty brine has actually been
sampled in locations where it issues onto the sea floor, and appears to be
derived from solution of rock salt (halite) and gypsum within strata under
Florida. The
quantity of ground water seepage into the ocean on a global scale can be
estimated from the quantity of global yearly rainfall minus global evaporation
as compared to global river runoff. Global river runoff is about 10% less than
global rainfall minus evaporation [19]. This missing water from the continents
(approximately 3.7 x 1015 L/yr) is
believed to be the total ground water seepage from the continents. An average
Na+ concentration for this seepage would allow the Na+ flux to the ocean to be
calculated. However, we know this average imperfectly. If we assume that the
average ground water has 26 mg/L of Na+ (5 times
the concentration of average river water), the global Na+ flux is A1O = 9.6 x
1019 kg/yr from ground water seepage. This rough estimate represents 50% of the
total river input. If further research should increase the global average Na+ content of ground water,
the flux of this
very significant source would increase. All. Sea-floor Hydrothermal Vents.
Fifteen years of intense investigations of hot springs on the deep ocean
floor have led geochemists to the understanding that large quantities of ocean
water are circulated through a significant fracture system in hot sea-floor
rocks. Some of the springs have water hotter than 350°C containing
significantly more total dissolved solids than seawater. This indicates
seawater alters sea-floor basalt by a complex series of metamorphic reactions.
According to Seyfried [20], metasomatism dissolves Na+ from basalt below 350°C but fixes Na+
in mineral phases above 350°C. Two classes of sea-floor hydrothermal vents are recognized
by Wolery and Sleep [21]: axial and off-axial hydrothermal vents. The axial
hydrothermal vents occur along the axes of mid-ocean ridges where active rift
faulting has brought the hottest basalts near the ocean floor. These springs
are dominated by water hotter than 250°C. The off-axial vents are located
on the flanks of the mid-ocean ridges away from the recent rift faulting. These
springs are dominated by water from 80 to 250°C, which is
cooler than the axial springs. Using data on latent heat and heat flow from
mid-ocean ridges, Wolery and Sleep [22] estimate the upper limit of water flux on a
global scale through mid-ocean ridges: 2.3 x
1013 kg/yr. They believe that 17% of
the water flows through axial vents and 83% flows through off-axial vents.
Chemical compositions of vent waters allowed Wolery and Sleep [23] to estimate
that the off-axial vents add 1.1 x 1010 kg of Na+
each year to the ocean. Estimation of the global
contribution of Na+
by way of the axial vents is complicated by the difficulty in measuring
the actual vent temperature and calculating the rock to water ratio in these
springs. Furthermore, two dozen measurements of axial springs may not be
representative of the global flow. Twenty axial springs from the
Pacific Ocean have an average Na+
concentration of 502 mmol/kg, showing an enrichment
of 38 mmol/kg
above normal seawater [24]. Three axial springs from the Atlantic Ocean have an
average Na+ concentration
of 534 mmol/kg,
showing an enrichment of 70 mmol/kg above normal seawater [25]. Therefore, the
axial spring data indicate that axial hot springs in the Pacific and Atlantic
Oceans are enriched by 42 mmol/kg in Na+. Using the global water
flux for axial vents of 3.8 x
1012 kg [22], the global Na+ flux from axial vents is 3.7 x 109 kg of Na+ added to the ocean each
year. Addition of the
calculated Na+ inputs
from off-axial and axial hydrothermal springs gives the global hydrothermal
vent flux: All = 1.5 x 1010 kg/yr. This is 8% of the
total river input. PRESENT OUTPUTS Of Na+ FROM THE OCEAN Let us define Bi as the mass per unit time of Na+ taken out of the ocean by the ith sink.
What follows below and in Table 2 is a compilation of the seven known or
conjectured natural Na+
outputs from the ocean. These outputs are those from the
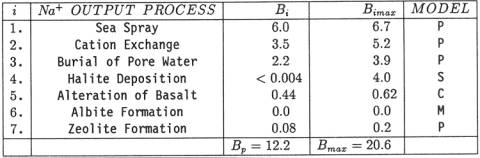
exhaustive list of Holland [26]. Table
2: Outputs of Na+
from the ocean. Units are in 1010 kg/yr. Present outputs are listed
in column headed Bi. Maximum past outputs are
listed in column headed Bimax. Models for estimating Bimax
are denoted "M" for "Modern Earth Model",
"P" for "Pleistocene Earth Model", "C" for
"Cretaceous Earth Model", and "S" for time averaged salt
deposits of the Permian System. B1. Sea
Spray. Waves of the sea, especially breaking waves along the shore, produce
air bubbles in the water. Collapse of these bubbles shoots into the air
droplets of seawater which evaporate to form microscopic crystals of halite.
Crystals of halite are carried with other aerosols by the winds from the ocean
to the continents. A major quantity of these aerosols form condensation nuclei
for clouds, some are scrubbed from the atmosphere by rain, and a small fraction
falls out dry onto the earth. Analyses of numerous rain water samples from five
continents by Meybeck [27] indicate that average rainwater has 0.55 mg/L of Na+. This average rain value
is probably in excess of the actual average because recent studies show that
Asia, the continent with the greatest rainfall, averages less than 0.4 mg/L
[28]. Furthermore, 2000 m of ice core from Antarctica, the earth's longest
duration aerosol record, averages less than 0.1 mg/L [29]. Using the value for
global precipitation over the continents of 1.10 x 1017 L/yr [30]
and Meybeck's generous value of 0.55 mg/L of Na+ in average rainfall, we obtain the mass of sea
spray deposits of Na+
on the continents: B1 = 6.0 x 1010kg/yr. This output is 31% of the total river input. B2. Ion Exchange.
Clays exhibit significant cation exchange ability especially in response to
changes in the chemical environment. River-borne clays have their cation
exchange sites dominated by Ca+2
because of the relatively high proportion of Ca+2 to Na+
in river water. However, at the mouth of rivers upon entering the ocean
these clays encounter seawater which has a significantly higher proportion of Na+ relative to Ca+2. As a result river-borne clays
release C a+2 from their cation exchange sites
and absorb Na+. The ability
of river-borne clays to absorb seawater Na+ is limited by the concentration of Ca+2 on the cation exchange sites. Sayles and Mangelsdorf
[31] have studied the cation exchange characteristics of clays of the Amazon
River, the world's largest river. Analysis of the river-borne clay of the
Amazon showed that the cation exchange process occurs very rapidly as the clays
enter the ocean. At the most frequently encountered discharge and sediment
levels of the Amazon, Sayles and Mangelsdorf estimate that 20% of the
river-born Na+ is
absorbed as clays enter the ocean. This was confirmed by laboratory experiments
on the cation exchange abilities of river-borne clay [32]. Using this data,
Drever, Li and Maynard [33] estimated the global uptake of Na+ by river-borne clays at B2 = 3.5 x 1010 kg/yr. This output is 18% of the
total river input. B3. Burial of Pore Water. Sediments
contain open spaces between their grains which in the ocean are filled with
pore fluids. Thus, there is some seawater lost each year from the ocean simply
by the permanent burial of pore water with the accumulation of sediments.
Drever, Li and Maynard [34] used the mass of ocean sediment added to the ocean
and accumulated on the sea floor annually (2 x
1013 kg/yr) and the average final porosity
(30%) to estimate the quantity of seawater removed. From the quantity of
seawater removed they calculate the flux of Na+ removed yearly by burial of pore water: B3 = 2.2 x 1010 kg/yr. This output is 11% of the total river input. B4. Halite Deposition. Many have assumed
that the major pathway for Na+
removal from today's ocean is the deposition of the mineral halite.
However, the major halite deposits accumulate currently from concentrated river
water on the continents, not from the ocean. Modern marine sedimentary deposits
are nearly devoid of halite. Recent marine salt flats and coastal lagoons occur
along the Persian Gulf, along the Gulf of California, and on the west coast of
Australia, but they have very meager deposits of halite. When halite is deposited
in marine salt flats and coastal lagoons, freshening of the brine after
deposition often redissolves the halite. Solution of halite in seawater occurs
because seawater is very undersaturated in both Na+ and Cl-. In fact seawater could contain 20 times
its present concentration of Na+
before deposition of halite would occur. Thus, modern sedimentary
conditions seem to prevent large, permanent accumulation of halite in marine
environments. The world inventory of modern marine halite deposits must be accumulating
today at a rate of less than 1 x 108 kg/yr. Thus, the flux of Na+ in modern marine halite
deposition is: B4 <
4 x 107 kg/yr. Today's oceanic output of
Na+ as
halite is trivial when compared to the modern river input. B5. Low Temperature Alteration
of Sea Floor Basalt. The coolest basalts which form the uppermost rock of
the ocean floor also circulate seawater but the temperatures of these fluids
usually remain below 60°C. At this lower temperature the basalt is weathered to
form clay minerals. Drilling of the upper 600 meters of oceanic basalt showed 5
to 15% weathering of basalt to form very pervasive clays [35]. The primary clay is saponite, a Na+-containing
mineral of the smectite (montmorillonite) group. Therefore, Na+
from low-temperature seawater reactions with the basalt must remove Na+
from seawater. The quantity of clays in basalt was reported by Wolery and Sleep
[36] to require removal of about 4.4 x 109 kg/yr of Na+.
Thus, removal of Na+ by low temperature alteration of basalt is B5 =
4.4 x 109 kg/yr. This output is 2% of the total river input. B6.
Albite Formation. Sea floor basalts above 350°C contain fluids
which exchange Na+ for Ca+2 [20]. This
metasomatic process, which occurs beneath the ocean, transforms calcium-rich
feldspars (anorthite) to Na+-rich feldspars (albite). Evidence of the process
is seen in chlorite-grade metamorphism from basalts dredged from the sea floor.
However, as discussed earlier in the input section, the axial hydrothermal
vents, even many of those which emit water over 350°C, generally show
enrichment, not depletion, of Na+ [37]. The suggestion by
Holland [38] that albite formation is an effective sink for oceanic Na+
is not supported by the most recent data. It appears that the seawater as it is
heated from ocean temperature to 350°C gains as much or more Na+ from low
temperature solution of Na+ in basalt as is removed above
350°C. Thus, there does not appear to be any significant removal of Na+
from the seawater by the formation of albite. The Na+ used
in albite formation appears to come from within the ocean crust. It is
concluded that albite formation removes essentially no Na+
from the ocean. Therefore, B6 = 0 kg/yr. B7. Zeolite Formation. Minerals of the
zeolite group are strong absorbers of alkalies (Na+, K+)
from seawater and are found in small amounts in ocean sediments. Phillipsite
and clinoptilolite, Na+-rich members of the zeolite group, form from alteration
of volcanic ash. According to Mackenzie and Wollast [39] about 4x1010
kg of volcanic ash are added to the ocean yearly. If fully one-half of this
volcanic ash (averaging 3% by weight Na+) is converted to
phillipsite (averaging 7% by weight Na+), 8 x 106
kg/yr of Na+ would be removed from seawater. Thus, a generous allowance for
zeolite formation suggests B7 = 8 x 108 kg/yr of Na+
removal. Holland [40] recognizes the removal of seawater Na+
by zeolites, but admits the quantity is minor. The output of Na+
calculated for zeolites is less than 1% of the total river input. EVOLUTIONARY
EARTH MODELS Constraints
on the minimum inputs and maximum outputs for Na+ can be
established by examining three different earth models. These are (1) the
Pleistocene Earth Model, (2) the Cretaceous Earth Model, and (3) the Modern
Earth Model. These models have been elaborated by evolutionists and are
employed here to evaluate the limits of Na+ variation in the
history of earth's dynamic systems. The
Pleistocene Earth Model (abbreviated "Model P") was generated by
geologic evidences of widespread continental glaciation. It supposes that the
earth experienced an "ice age" [41]. A large area of northern Europe,
Asia and North America was covered by continental glaciers when global mean
temperature was about 10°C. Compared to today's earth, sea level was
lower, about 5% greater area of continents was exposed, and there was greater
length of coastline. Total global rainfall was greater than today, and, because
of higher river discharge, more elevated continents, and much reduced desert
areas, global erosion was more rapid than today [41]. Volcanism was extensive
judging from the size and abundance of Pleistocene calderas, but rift faulting
at mid-ocean ridges was occurring near today's rate [42]. The
Cretaceous Earth Model (abbreviated "Model C") is based on fossil
flora and fauna from Cretaceous strata indicating that warm climate extended
into polar latitudes [43]. There are supposed to have been no glaciers and
global mean temperature may have exceeded 20°C [42]. Higher sea level
would have caused the area of Cretaceous continents to be 95% of today's
continents [44]. Global rainfall and global continental drainage by rivers may
have been 25% greater than today's [44], but because of the reduced elevation
of the continents, less continental area, and more extensive soil development,
the rate of erosion and sedimentation was about 54% of today's [44]. Cretaceous
sea-floor spreading has been supposed to have occurred at 1.4 times today's
rate [42]. Perhaps, because of more volatiles released by accelerated tectonics
on the sea floor, there was four times the present level of atmospheric C02
[42]. Chemical weathering would have been greater than today because of
increased soil humidity and acidity [42]. The
Modern Earth Model (abbreviated "Model M") is based on our recent
earth which is available for our direct study. Today's earth has 15°C
global mean temperature and, because of modern continental glaciers, more
closely resembles Model P than Model C. An important distinctive of our modern
earth is its aridity. Desert areas characterize large portions of our
continents which have reduced river discharge. MINIMUM
PAST Na+ INPUTS ACCORDING TO EVOLUTIONARY MODELS We
seek to determine minimum past input rates, Aimin for each of the
eleven processes delivering Na+ to the ocean. We
can use our earth models to make this evaluation for the purpose of discerning
whether evolutionary explanations for the earth's ocean allow the ocean's Na+
content to remain in steady state. Minimum values for eleven Na+
input processes (Aimin) are listed in Table 1. For the sake of
calculation, we assume the "steady state" condition where the ocean's
Na+ concentration does not change with time. The
flux of river Na+ from wash out of sea spray aerosol depends
on the length of shoreline, area of continents, energy of waves, and
concentration of Na+ in seawater. Assuming the steady-state
model (past Na+ concentration of seawater equivalent to
today's), we obtain the minimum sea spray river flux using Model C. Because
Cretaceous coastlines would be 97% of today's length and continental area for
aerosol to wash out would be 95% of today's, A1min = 0.97 x 0.95 x A1. In
a similar fashion past minimum global fluxes can be estimated for inputs A2
through A11. The rate of release of Na+ to rivers
by silicate weathering (input A2) is primarily dependent on soil
acidity and soil humidity [45]. Soil acid, which is produced primarily from C02
generated by organic decay, is the most effective agent for release of Na+
from silicate minerals. High soil humidity is the factor which increases
organic activity in soils, and, in addition, makes possible the leaching of Na+
from soils to rivers. Thus, it can be argued that Model M with modern, more arid
and alkaline soils would produce the minimum global flux of silicate-derived Na+
to the oceans through rivers. Models P and C have more humid and acidic soils
than Model M. The area of modern deserts (where low Na+
solution from silicates occurs) in Model M more than offsets the increased area
covered by glaciers (where low solution of Na+ occurred) in
Model P. Indeed, it is difficult to imagine an earth model where less Na+
is delivered by rivers to the ocean from weathering of silicates. Therefore, A2min
= A2. The
flux of Na+ through rivers from solution of chlorides on the
continent is related to global precipitation and to area of exposed chloride
deposits on the continents. All earth models suppose about the same area of
exposed continental chlorides, so Model M, the model with the most arid
climate, would have the lowest solution rate. Thus, A3min =A3. The
expulsion of Na+ from ocean sediments (A4) is
directly related to the rate of sedimentation, the lowest sedimentation rate
producing the lowest input of Na+ from buried sea-floor
clays. The lowest sedimentation rate is for Model C, evaluated at 54% of Model
M by Tardy et al. [44]. Thus, A4min = 0.54 x A4. For
inputs of Na+ by marine erosion (A7) and ground
water (A10), Model C gives the minimum Na+ inputs
because marine erosion and ground water fluxes are related most strongly to the
length of shorelines. Length of Cretaceous shorelines would be about 97% that
of modern shorelines. Thus, A7min = 0.97 x A7 and A10min
= 0.97 x A10. Sea-floor spreading has been regarded by evolutionists
to be slowing down with time [42]. Therefore, the lowest output of Na+
from sea-floor hydrothermal vents is today's: A11min= A11. MAXIMUM
PAST Na+ OUTPUTS ACCORDING TO EVOLUTIONARY MODELS We can also evaluate
the past outputs of Na+ from the ocean and estimate each Bimax
the maximum output values for each output process. These are listed in Table 2.
The quantity of Na+ removed from the ocean by the sea spray
process (B1) is, as stated before, related to length of shoreline,
area of the continents, energy of waves, and concentration of Na+
in seawater as each sea spray droplet formed. Evolutionists have supposed the Na+
concentration of seawater and salt spray droplets have remained roughly
constant over hundreds of millions of years. Thus, Model P with the most
shoreline, the greatest continental area, and the greatest wave energy produces
the greatest sea spray flux. A 2000 m deep ice core from Antarctica [29] contains old ice left over from the Pleistocene.
That ice, however, does not contain a significantly higher Na+
aerosol content than recent ice
deposited on Antarctica. Thus, past maximum rates of removal of Na+
by sea spray are only slightly
greater than modern rates. A generous allowance gives a good value, B1max = 6.7x1010 kg/yr. Approximately 1.5 times the present river sediment load would be carried to
the oceans with Model P [44]. This largest global load of sediment in an
evolutionary model would allow the largest Na+ exchange from seawater to river sediments and bury
the most pore water within ocean sediments. Thus, it can be estimated that B2max
= 5.2 x 1010 kg/yr and B3max = 3.9 x
1010 kg/yr. The most Na+
removal by alteration of ocean floor basalt would occur in Model C where
sea floor is supposed to form 1.4 times
faster than today. The value of B5max = 0.62 x
1010 is estimated. Evolutionists have claimed that the process of halite
deposition (B4)
is much different today than in the
past. They admit that modern marine halite deposits are of trivial volume, but
attribute ancient massive halite deposits to short, irregularly occurring
episodes. Drever, Li and Maynard speak for many evolutionists who believe:
"...such events appear to be well able to absorb the river excess over
long periods of time ...... [46]. Na+ in
earth's halite deposits is a relatively small sink for Na+, as can be appreciated by
"time averaging" it over the supposed duration of the deposits. The
present inventory of rock salt in the earth's strata contains about 4.4x
1018 kg of Na+
[47] which is 30% of the mass of Na+
in the ocean. Dividing the present mass of Na+ in global rock salt (4.4 x 1018 kg) by
the supposed duration of the Phanerozoic deposits (6
x 108 yr) gives an average rate of Na+ removal for the Phanerozoic of 7.3
x 109 kg/yr. This flux is an order of magnitude less than the
sea spray output process (B1.".) and cannot serve to balance during long
time intervals any of the major input processes (A1min,
A2min, A3min or A10min). Furthermore, it is extremely
unlikely that the "time averaged" halite output contains a
significant error. No major quantity of halite in the earth's crust could have
escaped our detection. Because halite is dominantly a basinal deposit on
continents, it is unlikely that any major quantity has been extracted by
subduction from the crust into the mantle. We can estimate B4max by an analysis of halite deposits of the Permian System.
The Permian contains the world's thickest and most extensive marine halite
deposits. Of the 4.4 x 1018 kg of Na+
in the earth's rock salt, 1.0 x 1018 kg (23%) resides in Permian rock salt [48]. Assuming that 50% of the Permian halite strata have survived erosion (a good
estimate based on the continental exposure of Permian basinal deposits), the
original Permian Na+
mass would be 2 x 1018 kg. The
"time averaged" maximum rate of removal of Na+ by halite deposition is
estimated in reference to the supposed 50 million year duration of the Permian Period. The maximum
rate of Na+ removal by
marine halite deposition (B4max)
is 4.0 x 1010 kg/yr [49]. The rate is only 67% of the present river input of Na+ derived weathering of silicates (A2). Even
more interesting is the observation that B4max is about half the present river flux derived from solution of
continental chloride minerals (A3). Past halite deposition (B4max) is not the major process that has been supposed: it ranks
third behind past sea spray (B1max) and cation exchange (B2max). Halite in the
earth has not been the major sink for Na+ generated by supposed hundreds of millions of years
of continental weathering. SIGNIFICANCE OF THE IMBALANCE Data that have been assembled in summary form in Tables 1
and 2 show the enormous imbalance of Na+ inputs compared to outputs. Ap, the
total of the eleven present Na+
inputs, is 4.57 x 1011 kg/yr,
whereas Bp, the total of the seven present outputs, is only 1.22
x 1011 kg/yr. The present
output to input ratio (xp = 0.27) shows that only 27% of Na+
going into today's ocean can be accounted for by known output processes.
If the "steady state" model is correct, xp should be equal
to 1.0, not 0.27! It is
extremely unlikely that one major or several minor Na+ output processes, comprising 3.35 x 1011 kg/yr of Na+,
have eluded our detection. That
the Na+ imbalance
exists in the ocean is further corroborated by consideration of CI-,
the primary anion which balances the charge of input Na+. According to Drever, Li
and Maynard [50], Cl- also being added to the ocean at a
much faster rate than it is being removed. Thus, we have strong evidence that
the ocean is not presently in "steady state" condition. If the
inputs of Na+ were constant in time and there were no
outputs, the time τ it would take to bring the mass of Na+
in the ocean from zero to today's amount Mp would
be: In a
similar fashion the maximum time required to bring the ocean to its present Na+
level can be calculated assuming the slowest possible Na+
input processes: Evolutionists
call τ the "residence time" of Na+, implying
that τ is the average time a Na+ ion spends
in the ocean. However, as already demonstrated, the present oceans are not in
steady state, so τ cannot be the "residence time" for Na+.
For clarity of concepts, we call τ the "filling time". Estimates
of the Na+ filling time ("residence
time") in the literature over the last century have varied between 260 Myr [3]
and 26 Myr
[51], generally getting smaller with time as more Na+ inputs
have been identified and measured more accurately. Before a filling time (τ) can be calculated, Mp, the
present mass of Na+ in the ocean needs to be determined. The
ocean's concentration of Na+ today is 10,760 mg/kg
[52]. The mass of the oceans is 1.37
x 1021 kg [30], allowing the Na+ mass in
the ocean to be calculated: Mp =
1.47 x 1019 kg. The total of the eleven Ai's listed
in the "present inputs" section is Ap =
4.57 x 1011 kg/yr. Substituting the last two values in equation 1 gives a filling
time of 32.2 Myr. Because the input fluxes were estimated
conservatively, we can say: T < 32.2 Myr. The maximum filling time
calculated using equation 2 gives Tmax = 41.3
Myr. Because the minimum input fluxes were estimated very conservatively,
we can say Tmax <41.3 Myr. ESTIMATING
THE OCEAN'S AGE It is
important to understand that τ is not the age of the ocean. To get an age
estimate, we need to account for three other factors: (1) the output rates, (2)
the past behavior of inputs and outputs, and (3) the initial amount of Na+.
Let us consider first the effect of output rates. The three major outputs are
aerosol removal by sea spray (B1), cation exchange with river clays (B2), and
burial of pore water in ocean sediments (B3). Together these three
removal paths account for 96% of the present Na+
removal from the ocean (see Table 2). However, the rates of Na+
removal by each of these three processes are dependent on the concentration of Na+
in seawater. Lower rates of removal for the three processes would be expected
in the past when seawater had a lower concentration of Na+
Thus, these output rates cannot be constant through time, but must be
proportional to [Na+](t), the Na+
concentration of the ocean at some past time t, and also proportional to M(t), the mass of Na+
in the ocean at time t. We can
express the rates as Bi(t) = biM(t), where
each coefficient bi is a
proportionality constant. Next,
let us consider outputs. If the sea has been increasing its Na+
content continually, then today's three major outputs (B1,
B2 and B3) must
have been smaller in the past. Thus, one cannot simply subtract today's output
rates from the input rates and use a form of equation 1 to get the age.
Instead, we must solve a differential equation giving the rate of change of M(t) in terms
of the input rates Ai and the output rates Bi(t) [53]: where we have defined B = Σbi and At = ΣAi. If At and B are constant with time,
the solution of equation 3 is: as one
can verify by substitution. Here M0 is the initial mass of Na+
in the sea. We can solve this equation for the time T it would take the mass of
Na+ in the ocean to reach the present level, Mp: where τ is the fillup time of equation 1 and x
is the output-to-input ratio: Equation 5 would give the age of the ocean if At and β had been constant, conditions
which undoubtedly do not apply. However, we will use equation 5 in the
following discussion to establish a maximum
age for the ocean. We can say by the evolutionary models discussed
previously that At, the sum
of the Na+ inputs
at any time t in the past, has always been greater than or equal to Amin = 3.56 x 1011 kg/yr, the sum of the eleven past
minimum input rates or the processes in Table 1.
Similarly, we can say that Bt, the sum of the Na+ outputs at any time t in the past, has always been less than or equal to Bmax = 2.06 x 1011 kg/yr, the sum of the seven past maximum output rates
for the processes in Table 2. The ratio of these two values, Bmax/Amin, gives us a maximum value, xmax,
for the output-to-input ratio: To make our age estimate as large as possible for the
benefit of the evolutionary model, we set "the initial Na+ mass M0= 0,
even though the creationist model would suggest otherwise. Then, we insert xmax from equation 7 and
τ max from equation
2 into equation 5 to get an expression for the absolute upper limit for the age
of the ocean: Using τ max
= 41.3 Myr and xmax
= 0.58 in equation 8 gives T < 62 Myr. OUTLINE OF A CREATIONIST MODEL To get a maximum age for the ocean according to an
evolutionary model, we had to assume zero initial Na+ in the sea, but there is no reason for the
creationist model to make such an assumption. On the contrary, there may be
good biological reasons to expect God to have created the original ocean with
significant salinity. In the maximum age calculation we also assumed an
evolutionary model with no catastrophic additions of Na+ to the ocean. The Genesis
Flood, however, would have added highly saline subterranean waters to the
oceans (the "fountains of the great deep", Genesis 7:11).
Furthermore, Na+
would have been released by reactions with hot basalt spreading out from
the resulting mid-ocean ridges, reactions with volcanic ash and basalt, and the
massive runoff of waters from the continents (Genesis 8:3-5). For thousands of
years after the Flood, the climate would have been hotter and wetter than
today, causing enhanced amounts of Na+
solution. Extensive post-Flood volcanoes would have deposited enormous quantities
of volcanic ash which would have weathered and delivered Na+ to the oceans at a much
higher rate than today. Thus, the creationist model implies (1) that the
initial level of Na+ in the ocean was a substantial fraction
of today's level, (2) that there was a significant burst of input Na+ during the Genesis Flood, and
(3) the Na+ input
rate was at higher levels than today for thousands of years. CONCLUSION Equation 8 reduces the
entire controversy down to one question: what is the value of xmax? Evolutionists and old-earth
creationists must assert that the ocean is in a steady state condition, meaning
that input and output rates have been about equal throughout geologic time, on
the average. By that view, they assert that xmax = 1. This means that T would be infinite, and we
could say nothing about the age of the ocean from its Na+ content. However, data we have
been able to compile from our knowledge of the earth, indicate that the present
output of Na+ from the
sea is only one-quarter the present rate of input (xp = 0.27). Furthermore, taking into
account plausible evolutionary earth models with maximum outputs and minimum
inputs we still cannot solve the dilemma. Our most generous output and input
models give xmax = 0.58.
This means (1) that the evolutionary steady-state model is inconsistent with
the data, and (2) that the ocean is much younger than the 3-billion year age
evolutionists commonly suppose. The data and equation 8 limit the ocean's age
to less than 62 million years. The
significance of this result is: (1) the evolutionary timescale of geologic
events associated with the ocean is grossly wrong in an absolute sense (though
not necessarily in a relative sense), (2) the corresponding radiometric dating
methods are grossly wrong (probably because of assumptions implicit in the
methods), and (3) biologic evolution, which is alleged to have started in the
ocean and had most of its history there, has not had time to occur. Our
result is an upper limit on the age of the ocean. It does not mean that the
true age is anywhere near 62 Myr.
According to the creationist model, most of the Na+ in the ocean is there as a result of Creation and
the Genesis Flood, not as a result of Na+ input due to geologic processes sustained over a billion
years. This leaves room for the possibility that the sea is less than ten
thousand years old. Our conclusion from the Na+ data is that the sea is less than 62 million years old. This is at least fifty times
younger than the age evolutionists require it to be. We
challenge evolutionists and old-earth creationists to report quantitative data
supporting a steady state ocean. Those who propose that continental weathering
and rivers have been delivering Na+
to the ocean for 3 billion years need to explain the sea's missing salt.
We urge Van Till, Young and Menninga to justify their assertion: "The 4.5
billion year chronology of earth history is in no way weakened or disqualified
by an appeal to the salt content of the terrestrial oceans" [7]. REFERENCES [1] Halley, E., "A short account of the cause of the
saltness of the ocean, and of the several lakes that emit no rivers; with a
proposal, by help thereof, to discover the age of the world,"
PHILOSOPHICAL TRANS. ROYAL SOC. LONDON, Vol. 29, 1715, pp. 296-300. [2] Joly, J., "An estimate of the geological age of
the earth," SCIENTIFIC TRANS. ROYAL DUBLIN SOC., New Series, Vol. 7, Part
3, 1899. Reprinted in ANNUAL REPORT SMITHSONIAN INSTITUTION, June 30, 1899, pp.
247-288. [3] Livingstone, D. A. "The sodium cycle and the age of the
ocean," GEOCHIM. COSMOCHIM. ACTA, Vol. 27, 1963, p. 1055. [4] Clarke, F. W.
"The data of geochemistry, 5th ed.," U. S. GEOLOGICAL SURVEY BULL.,
Vol. 70, 1924, 841 pp. Goldschmidt, V. M., "Grundlagen der quantitativen
geochemie", FORTSCHR. MINERAL. KRISTALLOGR. PETROGR., Vol. 17, 1933, pp.
1-112. Rubey, W. W., "Geologic history of sea water. An attempt to state
the problem", GEOL. SOC. AMER. BULL., Vol. 62, 1951, pp. 1111-1148. [5] Gregor, C. B., "Prologue: cyclic processes in
geology, a historical sketch", in Gregor, C. B., Garrets, R. M.,
Mackenzie, F. T., and Maynard, J. B., eds. GEOCHEMICAL CYCLES IN THE EVOLUTION
OF THE EARTH, John Wiley, New York, 1988, pp. 5-16. Quote from p. 13. [6] Van Till, H. J., Young, D. A., and Menninga, C.,
SCIENCE HELD HOSTAGE, Intervarsity Press, Downers Grove, IL., 1988, 189 pp.
Chapter 5, "Timeless Tales from the Salty Sea", critiques creationist
views of ocean salinity and affirms the steady state model. [7] Van Till, Young and Menninga, op. cit., p. 91. [8] Meybeck, M., "Concentrations des eaux fluviales
en elements majeurs et apports en solution aux oceans," REV. DE GEOL.
DYN. GEOGR. PHYS., Vol. 21, 1979, pp. 215-246. See tabulation of data in Tables
1 and 5. [9] Meybeck, op. cit., Table 6. [10] Meybeck, M., "Global chemical weathering of surficial rocks
estimated from river dissolved loads", AMER. JOUR. SCI., Vol. 287, 1987,
pp. 401-428. [I1]
Sayles, F. L., "The composition and diagenesis of interstitial solutions;
I. Fluxes across the seawater-sediment interface in the
Atlantic Ocean", GEOCHIM. COSMOCHIM. ACTA, Vol. 43, 1979, pp. 527-546.
Table 7, column 6 gives the fluxes of ions out of and into ocean sediments. [12]
Drever, J. I., Li, Y. H., and Maynard, J. B., "Geochemical cycles: the
continental crust and the oceans", in Gregor, C. B., Garrets, R. M.,
Mackenzie, F. T., and Maynard, J. B., eds., CHEMICAL CYCLES IN THE EVOLUTION OF
THE EARTH, John Wiley, New York, 1988, pp. 17-53. See Table 1.4, column 3. [13]
Schultz, D. F., and Turekian, K. K., "The investigation of the
geographical and vertical distribution of several trace elements in sea water
using neutron activation analysis", GEOCHIM. COSMOCHIM. ACTA, Vol. 29,
1965, pp. 259-313. [14] Garrels, R. M., and Mackenzie, F. T., EVOLUTION OF
SEDIMENTARY ROCKS, W. W. Norton, New York, 1971, Table 4.11. [15] Pye, K., AEOLIAN DUST, Academic Press, New York,
1987, p. 90. [16] Mackenzie, F. T., and Wollast, R., "Sedimentary
cycling models of global processes", in Goldberg, E. D., ed., THE SEA,
John Wiley, New York, Vol. 6, 1977, p. 742. (17] Meybeck, "Concentrations des eaux
fluviales...", Table 6. (18] Paull, C. K., and Neumann, A. C., "Continental
margin brine seeps: Their geological consequences," GEOLOGY, Vol. 15, June
1987, pp. 545-548. [19] Garrels and Mackenzie, EVOLUTION OF SEDIMENTARY
ROCKS, p. 104. [20] Seyfried, W. E., Jr., "Experimental and theoretical constraints
on hydrothermal alteration processes at mid-ocean ridges", ANN. REV. EARTH
PLANET. SCI., Vol. 15, 1987, p. 324. [21]
Wolery, T. J., and Sleep, N. H., "Interactions of geochemical cycles with
the mantle," in Gregor, C. B., Garrels, R. M., Mackenzie, F. T., and
Maynard, J. B., eds., CHEMICAL CYCLES IN THE EVOLUTION OF THE EARTH, John
Wiley, New York, 1988, pp. 77-103. (22] Wolery and Sleep, op. cit., p. 91. [23] Wolery and Sleep, op. cit., table 3.5. [24] Von
Damm, K. L., "Systematics of and postulated controls on submarine
hydrothermal solution chemistry", JOUR. GEOPHYS. RES., Vol. 93, 1988, pp.
4551-4561. See Table 1 for Na+ concentrations of 20 Pacific hydrothermal
solutions. [25] Campbell, A. C., et al., "Chemistry of hot
springs on the Mid-Atlantic Ridge," NATURE, Vol. 335, (6 Oct. 1988), pp. 514-519. [26] Holland, H. D., THE CHEMISTRY OF THE ATMOSPHERE AND
OCEANS, John Wiley, New York, 1978, 351 pp. See table 5.14. [27] Meybeck, "Concentrations des eaux
fluviales...", p. 242. [28]
Petrenchuk, 0. P., "On the budget of sea salts and sulfur in the atmosphere",
JOUR. GEOPHYS. RES., Vol. 85, 1980, pp. 7439-7444. Petrenchuk, 0. P., and
Selezneva, E. S., "Chemical composition of precipitation in regions of the
Soviet Union", JOUR. GEOPHYS. RES., Vol. 75, 1970, pp. 3629-3634. [29] De Andelis, M., Barkov, N. I., and Petrov, V. N.,
"Aerosol concentrations over the last climatic cycle (160 kyr) from an
Antarctic ice core", NATURE, Vol. 325, 1987, pp. 318-321. [30] Berner, E. K., and Berner, R. A., THE GLOBAL WATER
CYCLE: GEOCHEMISTRY AND ENVIRONMENT, Prentice-Hall, Englewood Cliffs, New Jersey, 1987, 387 pp. [31]
Sayles, F. T., and Mangelsdorf, P. C., "Cation- exchange characteristics
of Amazon River suspended sediment and its reaction with seawater",
GEOCHIM. COSMOCHIM. ACTA, Vol. 43, 1979, pp. 767-779. [32] Sayles,
F. T., and Mangelsdorf, P. C., "The equilibration of clay minerals with
seawater: exchange reactions", GEOCHIM. COSMOCHIM. ACTA, Vol. 41, 1977,
pp. 951-960. [33] Drever, Li and Maynard, op. cit., Table 1.3,
column 3. [34] Drever, Li and Maynard, op. cit., p. 27. [35]
Muehlenbachs, K., "The alteration and aging of the basaltic layer of sea
floor, oxygen isotopic evidence from OSPDP/IPOD legs 51, 52, and 53",
INITIAL REP. DEEP-SEA DRILL. PROD., Vol. 51, 1980, pp. 1159-1167. [36] Wolery and Sleep, op. cit., Table 3.2. [37] Von Damn, op. cit.; Campbell et al., op. cit. [38] Holland, op. cit., Table 5.14, p. 232. [39] Mackenzie and Wollast, op. cit. [40] Holland, op. cit., p. 186. [41] Flint, R. F., GLACIAL AND QUATERNARY GEOLOGY, John
Wiley, New York, 1971, 982 pp. [42]
Berner, R. A., Lasaga, A. C., and Garrels, R. M., "The carbonate-silicate
geochemical cycle and its effect on atmospheric carbon dioxide over the past
100 million years", AMER. JOUR. SCI., Vol. 283, 1983, pp. 641-683. [43]
Barron. E. J., Thompson, S. L., and Schneider, S. H., "An ice-free
Cretaceous? Results from climate model simulations", SCIENCE, Vol. 212,
1981, pp. 501-508. [44]
Tardy, Y., N'Kounkou, R., and Probst, J., "The global water cycle and
continental erosion during Phanerozoic time (570 my)", AMER. JOUR. SCI.,
Vol. 289, 1989, pp. 455-483. See Table 7. [45]
Berner, R. A., and Barron, E. J., "Factors affecting atmospheric CO2
and temperature over the past 100 million years", AMER. JOUR. SCI.,
Vol. 284, 1984, pp. 1183-1192. [46] Drever, Li and Maynard. op. cit., p. 51. [47]
Estimate of global salt inventory by W. T. Hosler is cited by Holland, H. D.,
THE CHEMICAL EVOLUTION OF THE ATMOSPHERE AND THE OCEANS, Princeton Univ. Press,
Princeton, N.J., 1984, p. 461. [48]
Zharkov, M. A., HISTORY OF PALEOZOIC SALT ACCUMULATION, Springer-Verlag, New
York, 1981, 308 pp. Tables 7 and 8 contain data on volume of evaporites. [49] A
greater value for B4max can be
obtained if the very unusual Messinian (Late Miocene) evaporites of the
Mediterranean region are assumed to be of marine origin. W. T. Hosler et al.
("A census of evaporites and its implications for oceanic
geochemistry", GEOL. SOC. AMER, ABSTR. PROGRAMS, Vol. 12, 1980, p. 449)
estimate the Messinian rock salt mass, which allows the Na+ mass to be estimated at 5.8 x 1017 kg. This mass is about 13%
of the world's rock salt No+ and 4% of
the Na+ in the present ocean. If the
"Messinian salinity crisis" is assumed to have had a duration of one
million years, B4max would be 5.8 x 1011 kg/yr, a value
slightly greater than all the combined inputs. The "Messinian salinity
crisis", however, is admitted by many to be a truly extraordinary event.
It cannot be used to estimate the long term removal rate of Na+ in halite. [50] Drever, Li and Maynard, op. cit., p. 37. [51]
Billo, S. M., "Residence times of chemical elements in geochemistry",
AMER. ASSOC. PETROL. GEOL. BULL., Vol. 73, 1989, p. 1147. [52]
Holland, THE CHEMISTRY OF THE ATMOSPHERE AND OCEANS, op. cit., Table 5.1. [53] Lasaga, A.C., "The kinetic
treatment of geochemical cycles", GEOCHIM. COSMOCHIM. ACTA, Vol. 44, 1980, pp. 815-828. DISCUSSION The
world that we live in is God's world. We are reminded by many passages of
Scripture, and especially by Psalm 19:1 that "The heavens are telling the
glory of God; the skies proclaim the work of his hands." Therefore, we
must give serious consideration to what we learn about God's world through
scientific study of that world. From within that perspective, I submit the following
comments: 1) Our knowledge and understanding of
God's world is less than perfect and less than complete. We have not yet
learned in detail all of the processes involving sodium in the ocean
environment. Any improvements in our understanding are welcome. 2) The authors speak of the inability to
account for all of the factors which affect sodium concentration in the oceans
as a "dilemma" for those who think that the Earth is old. However,
even if the residence time of sodium in the oceans is less than the 260 million
years reported in much of the recent literature, we must remember that the
residence time of sodium in the oceans is not the same as the age of the Earth.
After all, the residence time of aluminum in the oceans is only 100 years, and
that is not viewed as a dilemma. 3) While short residence times for various
elements in ocean water are no dilemma for those who think the Earth is old,
long residence times for some elements in ocean water decidedly presents a
dilemma to those who think that the Earth is young. 4) Sodium is not the only element with a
residence time in the oceans which is longer that several thousand years. The
residence time for potassium is 11 million years, for magnesium is 45 million years,
for silver is 2 million years, and for uranium is 500,000 years. Are all of
those long residence times in error? Can all of them be reduced to several
thousand years by good data and proper calculations? 5) According to their own calculations,
the authors have determined a "filling time" of the oceans of 32.2
million years. Are the authors willing to accept that number as a minimum age
of the Earth? 6) After gathering a considerable amount
of data, and after performing several calculations and logical analyses, the
authors suggest that the scientific study they have done isn't worth anything,
after all. They suggest that God might have made the oceans recently, with
a great deal of sodium (and other elements) already dissolved in the water. If
that is the attitude one wishes to adopt, what is the justification for doing
the scientific study? A paper which is only one or two sentences in length
would suffice to reach the same result. 7) It is God's world that we are studying
by scientific methods. It is God's handiwork that we are learning about through
those studies. God deserves to be praised and honored--and believed--for what
we have been able to learn about his world. If our careful study of God's world
brings us false or unreliable information, then what can it mean for the
Psalmist to sing, "The heavens are telling the glory of God; the skies
proclaim the work of his hands."? Clarence
Menninga, Ph.D. Grand Rapids, Michigan CLOSURE We
had hoped Dr. Menninga would respond to our challenge "to report
quantitative data supporting a steady state ocean," but his review
contains no such data. An erroneous assumption underlies all of his remarks. It
appears implicitly in his points 2 through 4, and explicitly in point 5: he
assumes that residence times are minimum ages for the ocean, i.e., that the
ocean must be older than any given residence time. He does not explain his
reasoning, but it must be something like this: (1) If there were no initial
sodium (for example) in the ocean, and (2) if the input of sodium has always
been no greater than the present rate, then it would take more than 32 million
years (our residence time for sodium) to get the present amount of sodium in
the ocean. In other words, Dr. Menninga assumes a uniformitarian view of the origin
of sodium in the ocean; he feels that all the sodium in the sea got there by
today's processes at essentially today's rates. The
flaw in Dr. Menninga's reasoning is in his two uniformitarian "if"
conditions; he has no logical basis for assuming either is true. The
creationist model we described provides a specific counter-example; Menninga
cannot logically exclude the possibilities that (1) God created the ocean with
some initial sodium, and (2) the sodium input during the Flood was much higher
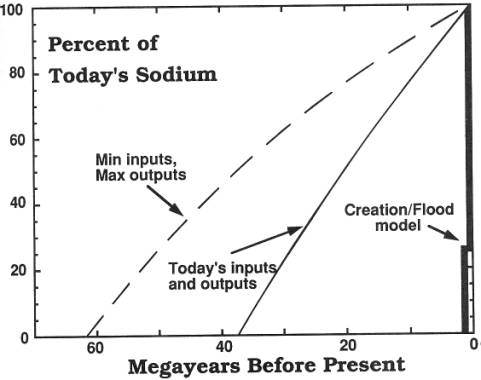
than it is today, a very natural consequence of such an event. (See Fig. A.) This shows that Menninga's
assumptions are not generally valid, so residence times are not minimum ages.
Our specific replies follow: 1.
"Our knowledge ... is less than perfect." This is basically an appeal
to unknown factors to support his view. "Improvements in our understanding
are welcome." Since our paper is the only one which has collected all the
diverse data on sodium inputs (including seven previously unrecognized ones) and
outputs, it should have improved Dr. Menninga's understanding. 2a.
"Inability to account for all of the factors." It is not we who profess
such an inability; we wrote that we have accounted for all of the major
factors. For over half a century, many evolutionists have been diligently
searching for sodium outputs, so we think it likely that all of the major ones
have been found. The dilemma for evolutionists is not in accounting, but in
facing up to the bottom line of the ledger: the sea is young. 2b.
"Residence time ... is not the same as the age." We agree; we never said
otherwise. Dr. Menninga evidently overlooked our statement stressing that
point: "It is important to understand that t [the residence time] is not
the age of the ocean." Apparently he also overlooked our main point, which
we emphasized numerous times in the paper: we have determined a maximum limit
on the age, not the age itself. Equation (8) specifies this limit, which
depends not only on the residence time but also on the maximum output-to-input
ratio, xmax. 2c.
Aluminum's small residence time is not a dilemma for old-earthers. Hence, he implies, sodium's
residence time should not present a dilemma, either. But it is not the
residence time which makes the dilemma; it is the imbalance between sodium
inputs and outputs. We can see this by contrasting what eq. (8) says about
aluminum and sodium. The data for aluminum gives xmax = 1; using this in eq.
(8) tells us that the age of the ocean is equal to or less than infinity. For
the data we report concerning sodium, eq. (8) tells us that the ocean is less
than 62 million years old. Both statements are true, but the one based on
sodium is more stringent, and that is the one which places evolutionists in a
dilemma. 3.
"Long residence times ... present a dilemma to [young-earthers]." This
would be true only if residence times were minimum ages, an idea we disproved
in our introductory remarks above. 4.
Can [various large residence times] be reduced to thousands of years? Since residence times are
not minimum ages, we are under no obligation to perform such a shrinkage. 5.
"Are the authors willing to accept [their 32 million year sodium residence
time] as a minimum age?" No. We can be persuaded by valid reasoning, but not
by mere repetition of the same error which underlies the previous points. 6.
"The authors suggest [their analysis] isn't worth anything." This suggestion comes from Dr. Menninga, not from us.
"What is the justification for the study?" The reason for our study
is the pursuit of truth. In the best tradition of science, the study rigorously
tests a hypothesis (the evolutionary view of the ocean), and it outlines a
testable alternative hypothesis, our creationist model. For reasons he does not
specify, Menninga disdains our model, but if he had paid close attention to its
implications, he might have recognized the flaws in his own argument. 7.
"God's world brings us false or unreliable information." Dr. Menninga's reaction to
our paper suggests that it is he who regards information from the natural world
as unreliable. He is avoiding a straightforward understanding of the sodium
data, because it does not fit into his preconceptions of an old earth.
"God's world ... deserves to be ... believed." So why doesn't Dr.
Menninga believe it? CONCLUSION Dr.
Menninga has staked a great deal upon his assumption that residence times
represent minimum ages; it is probably one reason he does not respond to our
challenge. He failed to see that our analysis and alternative model expose the
logical fallacies behind his assumptions and collapse his case. He also
misunderstood the thrust of the paper. Our main purpose was not to reduce the
residence time of sodium, but to quantify the gross imbalance between sodium
inputs and outputs and to clarify its implications. His response fails to make
crucial distinctions between four different concepts: residence time, maximum
age, minimum age, and true age. His decided preference for the term
"residence time" instead of the more neutral term "filling
time" clouds the central issue: Is ocean sodium in a steady state? Dr.
Menninga's repeated references to God and the Bible seem inconsistent with his
aversion to our creationist model. After all, the two main features of the
model came directly from scripture: (1) a recent creation, and (2) a worldwide
flood whose natural consequence would be a massive influx of sodium into the
ocean. We were aware that Menninga and his colleagues resist a straightforward
understanding of the Bible with regard to the youth of the earth. Evidently,
they similarly resist the biblical account of the Flood. We
are genuinely disappointed that Dr. Menninga did not overcome his
uniformitarian presuppositions enough to follow our reasoning clearly. We did
not expect agreement, but we did expect understanding. Therefore we call upon
Dr. Clarence Menninga and his colleagues, Drs. Davis Young and Howard Van Till,
to re-examine their presuppositions, read our paper more carefully, and respond
to our challenge: report quantitative data supporting a steady-state ocean. If
they cannot provide such data, then they should cease denying what we are
asserting: that all present knowledge about sodium in the sea indicates that
the ocean is young. Return To TCCSA Visitors
Pittsburgh, 1990, vol.2, pp. 17 - 33.
USED BY PERMISSION OF
CREATION SCIENCE FELLOWSHIP E-MAIL csf{at}trfn.clpgh.org
PO BOX 99303, PITTSBURGH, PA 15233
COPIES OF
THE PROCEEDINGS OF THE INTERNATIONAL CONFERENCES ON CREATIONISM
ARE AVAILABLE UPON REQUEST.
See their website http://csfpittsburgh.org
CREATION SCIENCE FELLOWSHIP is planning a 2003 International Conference and
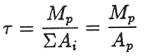
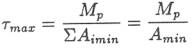
![]()
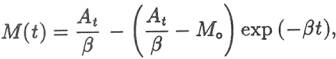
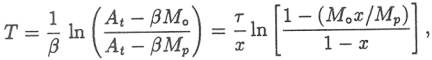
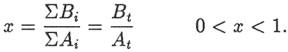
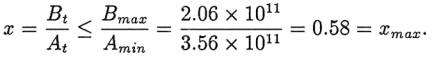
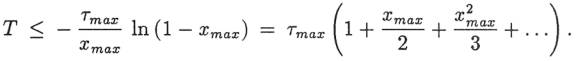
From "Proceedings of the Second International Conference on
Creationism,"
Pittsburgh, 1990, vol.2, pp. 17 - 33.
USED BY PERMISSION OF
CREATION SCIENCE FELLOWSHIP E-MAIL csf{at}trfn.clpgh.org
PO BOX 99303, PITTSBURGH, PA 15233
COPIES OF
THE PROCEEDINGS OF THE INTERNATIONAL CONFERENCES ON CREATIONISM
ARE AVAILABLE UPON REQUEST.
See their website http://csfpittsburgh.org
CREATION SCIENCE FELLOWSHIP is planning a 2003 International Conference and
calling for papers.